CAR T-cell Therapy in Cancer: Mechanisms, Challenges, and Prospects
By Jisara Wuttikreekiat | YSI Medicine
Cancer is one of the global causes of death that currently has no cure, with minimal therapeutic strategies. In 2017, the FDA approved CAR T-cell therapy, a CRISPR-made cancer therapy that has emerged as a groundbreaking approach in cancer treatment. It is a type of cancer immunotherapy treatment in which the patient’s immune cells (T cells) are genetically altered, enabling them to locate and destroy cancer cells more effectively. A particular CAR T-cell therapy, known as autologous CAR T cell therapy, begins by collecting the T cells from the patient’s blood using an apheresis machine, engineering the T cells in the lab, resulting in the manufacture of CAR T cells, and growing them before infusing them back into the patient. This essay will provide an overview of the mechanisms, challenges, and prospects of CAR T cell therapy in cancer.
Under normal circumstances, when a foreign substance is present in the body, the immune system recognizes it by finding proteins, called antigens, on the surface of those cells. Immune cells, called killer T cells, have receptors that recognize foreign antigens and help trigger other parts of the immune system to destroy the foreign substance. The same logic applies to cancer cells; however, as time goes on and mutations occur, cancer cells can develop genetic changes that help them escape the immune system. One way cancer cells escape the immune system is by avoiding recognition by killer T cells by reducing their number of MHC-I molecules (a molecule on the cell surface that presents short peptide fragments to T cells) on their surface. As a result of the reduction of MHC-I molecules, cancer cells are more likely to avoid being detected and eliminated by killer T cells, allowing them to grow and multiply uncontrollably.
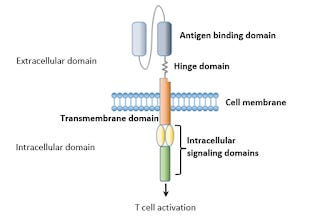
As scientists ponder the possibility of educating the patient’s T cells into better recognizing and attacking cancer cells, the idea of genetically modifying T cells arises. These modified T cells are referred to as Chimeric Antigen Receptor T cells (CARs), which are recombinants of receptors that help T cells and other immune cells recognize and attack specific cells. CARs are generally composed of four regions: the extracellular domain (antigen recognition domain and hinge region), a transmembrane domain, and the intracellular domain. The antigen recognition site recognizes antigens and interacts with the target antigen outside the cell; the domain is derived from a recombinant single-chain variable fragment, or ScFv, providing the function of target recognition. The hinge region, also known as the spacer, provides flexibility between the ScFv and the transmembrane domain, helping the ScFv recognize and bind to specific antigens on target cells. The transmembrane domain is a structural link between the extracellular domain and intracellular domain of the CARs; its function is to stabilize the receptor through a hydrophobic alpha-helix structure. Within the intracellular domain is the intracellular T cell signaling domain, which is derived from a costimulatory receptor such that when an antigen binds to the antigen recognition region, receptors come close and cluster together, resulting in the transmission of an activation signal inside the cell. The receptors are engineered to target specific tumor antigens independent of MHC antigen presentation, and once activated, they release cytotoxic compounds like perforin to kill the target cell.
While CARs hold the theoretical promise of curing cancer, in their therapeutic application, they are confronted with several challenges. According to Stanford Medicine, 9 in 10 CAR T-cell patients have cytokine release syndrome, as when CAR T cells multiply, they release chemicals called cytokines into the blood, which can ramp up the immune system. Its side effects include fever, headache, trouble breathing, low energy, etc. Although this syndrome would normally last a week, it is treatable with medication. However, there are more serious side effects to this therapy too; this includes allergic reactions during the infusion, low blood cell counts, temporary memory loss (lasting for a few days), etc. Other limitations to CAR T-cell therapy include the possibility of tumor antigen loss, which occurs when tumor cells downregulate or eliminate the target antigen, preventing CAR T-cells from recognizing them. Another possible limitation is that when T-cells are exposed to tumor antigens for prolonged periods, it can lead to T-cell exhaustion, causing CAR T-cells to be functionally impaired. Due to the nature of cancer cells’ mutations, it is extremely difficult for scientists to overcome these challenges and come up with a definite cure for various cancers. As different types of cancer have different antigens, specific CAR T cell therapy has to be made for each type of cancer.
Even though the effects of CAR T cell therapy are already challenging for patients, another pressing issue is its accessibility and cost. According to the College of American Pathologists, out of all the patients who could benefit from CAR T-cell therapy, only 20% have access to the treatment. As only about 150 hospitals in the US provide CAR T-cell therapy, this means that patients are required to travel miles in order to receive the treatment. This does not even account for the cost of the treatment, which is estimated to be approximately $500,000 to $1,000,000. Although there are ways to reduce the cost, not every family would be able to afford the treatment, and even if they have the money for the treatment, not only are they at risk, but they would also have to wait for the manufacturing process for approximately three weeks due to the autologous nature of the therapy.
In order for scientists to be able to save time in manufacturing the CAR T-cells for each individual, they are developing an allogeneic cellular therapy where the T-cells are obtained from healthy donor cells rather than from patients. With this allogeneic approach, it will enable rapid and scalable production of CAR T-cells as we eliminate the need to modify the patient’s T-cells. Moreover, this also means that CAR T-cells can be stored and used for multiple patients, ultimately reducing manufacturing costs. However, as there is not enough research, there are possibilities of immune rejection and off-target effects.
As of 2023, the FDA has approved six commercial CAR T-cell therapies, all of which are blood cancers. The complexity of targeting solid tumors with this therapy has presented significant challenges, thereby limiting the therapy primarily to hematologic malignancies. Nevertheless, researchers remain optimistic and are actively exploring the potential of CAR T-cell therapies not only for cancer, but also for autoimmune diseases. However, this research will take decades to perfect and it should proceed with caution due to the possible risks it poses as the therapy directly affects an individual’s DNA with slight mistakes that could potentially lead to one’s death. In conclusion, CAR T-cell therapy represents a groundbreaking advancement in the field of cancer treatment, but currently, there are too many risks involved with the treatment that it cannot be normalized as a treatment and needs further research to refine the techniques and ensure the patient’s safety.
References
Abramson Cancer Center. (n.d.). CAR T Cell Therapy. Pennmedicine.org. Retrieved December 30, 2023, from https://www.pennmedicine.org/cancer/navigating-cancer-care/treatment-types/immunotherapy/what-is-car-t-therapy
Ahmad, U., Khan, Z., Ualiyeva, D., Amissah, O. B., Noor, Z., Khan, A., Zaman, N., Khan, M., Khan, A., & Ali, B. (2022). Chimeric antigen receptor T cell structure, its manufacturing, and related toxicities; A comprehensive review. Advances in Cancer Biology - Metastasis, 4(100035), 100035. https://doi.org/10.1016/j.adcanc.2022.100035
American Cancer Society. (2022, March 1). CAR T-cell therapy and its side effects. Cancer.org. https://www.cancer.org/cancer/managing-cancer/treatment-types/immunotherapy/car-t-cell1.html
Beer, G. (2019, February 28). Science Surgery: ‘Why doesn’t the immune system attack cancer cells?’ Cancer Research UK - Cancer News. https://news.cancerresearchuk.org/2019/02/28/science-surgery-why-doesnt-the-immune-system-attack-cancer-cells/
Charrot, S., & Hallam, S. (2019). CAR-T cells: Future perspectives. HemaSphere, 3(2), e188. https://doi.org/10.1097/hs9.0000000000000188
Dagar, G., Gupta, A., Masoodi, T., Nisar, S., Merhi, M., Hashem, S., Chauhan, R., Dagar, M., Mirza, S., Bagga, P., Kumar, R., Akil, A. S. A.-S., Macha, M. A., Haris, M., Uddin, S., Singh, M., & Bhat, A. A. (2023). Harnessing the potential of CAR-T cell therapy: progress, challenges, and future directions in hematological and solid tumor treatments. Journal of Translational Medicine, 21(1). https://doi.org/10.1186/s12967-023-04292-3
ISB President Dr. Jim Heath and colleagues. (2022, November 10). For first time, scientists use CRISPR to treat cancer. Youtube.
Kim, S. K., & Cho, S. W. (2022). The evasion mechanisms of cancer immunity and drug intervention in the tumor microenvironment. Frontiers in Pharmacology, 13. https://doi.org/10.3389/fphar.2022.868695
Lumen Learning, & OpenStax. (n.d.). The adaptive immune response: T lymphocytes and their functional types. Lumenlearning.com. Retrieved March 21, 2024, from https://courses.lumenlearning.com/suny-ap2/chapter/the-adaptive-immune-response-t-lymphocytes-and-their-functional-types/
Memorial, B. (2021, January 27). Your immune system and cancer: What’s the connection? Beaufort Memorial Hospital. https://www.bmhsc.org/blog/your-immune-system-and-cancer-whats-the-connection
Mullin, E. (2023, November 29). A cutting-edge cancer treatment may cause cancer. The FDA is investigating. Wired. https://www.wired.com/story/a-cutting-edge-cancer-treatment-is-under-fda-investigation-for-cancer-risk/
NCI Staff. (2020, July 27). How CRISPR is changing cancer research and treatment. National Cancer Institute. https://www.cancer.gov/news-events/cancer-currents-blog/2020/crispr-cancer-research-treatment
Phillips, C. (2023, September 27). One CAR T-cell therapy for blood cancers? National Cancer Institute. https://www.cancer.gov/news-events/cancer-currents-blog/2023/universal-car-t-cell-blood-cancer
Robinson, K. M. (2023, January 24). CAR T-cell therapy: Managing costs and finding financial assistance. WebMD. https://www.webmd.com/cancer/lymphoma/features/navigate-finances-car-t-cell-therapy
Salik, J. (2022, September 2). Lymphoma: Overview of chimeric antigen receptor (CAR) T cells. Youtube.
Sheekey, E. (2023, October 21). Can we cure cancer with CRISPR? Youtube.
Singh, A. (2019a, January 17). Immunotherapy & CAR T Cells: Part 1. Youtube.
Singh, A. (2019b, January 17). Immunotherapy & CAR T Cells: Part 2. Youtube.
Snyder, S., Chung, K. C., Jun, M. P., & Gitlin, M. (2021). Access to chimeric antigen receptor T cell therapy for diffuse large B cell lymphoma. Advances in Therapy, 38(9), 4659–4674. https://doi.org/10.1007/s12325-021-01838-z
Stanford Health Care. (2019, May 29). Stanfordhealthcare.org. https://stanfordhealthcare.org/medical-treatments/c/car-t-cell-therapy/side-effects.html
Sterner, R. C., & Sterner, R. M. (2021). CAR-T cell therapy: current limitations and potential strategies. Blood Cancer Journal, 11(4), 1–11. https://doi.org/10.1038/s41408-021-00459-7
The Peter MacCallum Cancer Centre. (2022, October 5). CAR-T cells: Engineered cancer killers. Youtube.
Zanfagnin, V. (2022, November 30). CAR T-cells therapies: Opportunities and challenges. College of American Pathologists. https://www.cap.org/member-resources/articles/car-t-cells-therapies-opportunities-and-challenges
Zhang, Chen, Durer, S., Thandra, K. C., & Kasi, A. (2022). Chimeric antigen receptor T-cell therapy. StatPearls Publishing.
Zhang, Cheng, Liu, J., Zhong, J. F., & Zhang, X. (2017). Engineering CAR-T cells. Biomarker Research, 5(1). https://doi.org/10.1186/s40364-017-0102-y